News
- Home
- Medical Imaging QA Best Practices | QUBYX
Medical Imaging QA Best Practices | QUBYX
- March 1, 2026
- QUBYX Team
Medical Imaging QA Best Practices | A Structured Framework for Compliance, Accuracy, and Long-Term Performance
Why Medical Imaging QA Best Practices Matter
Medical imaging QA best practices are not optional technical enhancements — they are operational safeguards that protect diagnostic integrity, regulatory compliance, and institutional reputation.
In radiology, mammography, pathology, and enterprise imaging networks, diagnostic decisions depend on consistent grayscale performance, luminance accuracy, and controlled environmental conditions. Without a structured quality assurance (QA) framework, calibration becomes a one-time event instead of a continuous compliance process.
This guide outlines medical imaging QA best practices aligned with international standards and demonstrates how structured QA systems such as QUBYX PerfectLum transform calibration into an integrated governance model.
1. Align with International Standards
Effective medical imaging QA best practices begin with adherence to recognized technical frameworks:
-
DICOM Part 14 (GSDF) – Grayscale Standard Display Function
-
AAPM TG-270 – Acceptance and constancy testing for displays
-
DIN 6868-157 (Germany)
-
MQSA (Mammography Quality Standards Act, USA)
-
ACR technical standards
Compliance ensures:
-
Consistent grayscale rendering
-
Standardized luminance response
-
Reproducible diagnostic viewing conditions
Medical imaging QA best practices require calibration that aligns displays precisely to DICOM GSDF and verifies that performance remains within tolerance over time.
2. Implement Acceptance Testing at Deployment
Before clinical use, displays must undergo structured acceptance testing. This includes:
-
Maximum luminance (Lmax) verification
-
Minimum luminance (Lmin) measurement
-
Luminance ratio assessment
-
Uniformity testing
-
Ambient light evaluation
-
DICOM GSDF conformance
Acceptance testing validates that the device meets clinical and regulatory requirements before it enters workflow.
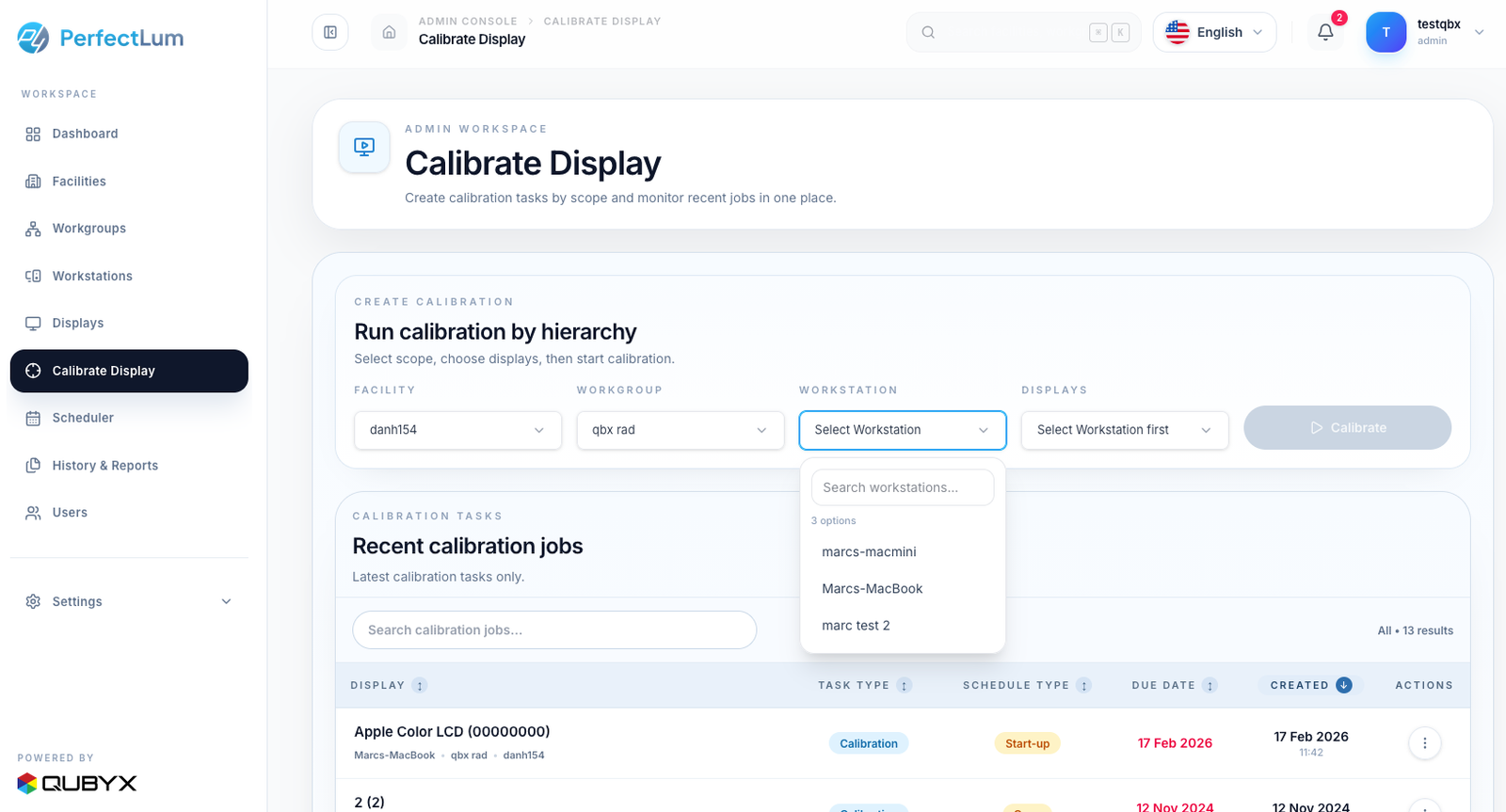
Using QUBYX PerfectLum, institutions can automate TG-270 aligned acceptance protocols and generate defensible baseline documentation.
3. Transition from Calibration to Continuous Monitoring
One of the most overlooked medical imaging QA best practices is recognizing that:
Calibration without monitoring is incomplete.
Displays drift over time due to:
-
Backlight aging
-
Panel degradation
-
Environmental changes
-
Hardware inconsistencies
Best practice requires:
-
Scheduled constancy testing
-
Automated luminance drift detection
-
Alert-based tolerance monitoring
-
Longitudinal performance tracking
QA must be continuous, not episodic.
4. Centralize QA Across the Enterprise
Modern imaging environments are no longer single-room setups. Hospitals and teleradiology networks often operate:
-
Hundreds of diagnostic displays
-
Multiple sites
-
Remote reading stations
-
OEM-integrated workstations
Medical imaging QA best practices recommend centralized governance.
A structured QA server architecture enables:
-
Global device inventory tracking
-
Role-based access control
-
Automated test scheduling
-
Centralized documentation storage
-
Audit-ready reporting
Enterprise-wide oversight ensures that no diagnostic display operates outside compliance thresholds.
5. Maintain Audit-Ready Documentation
Regulators and accreditation bodies require documentation that proves compliance — not assumptions of compliance.
Best practices include:
-
Full historical test logs
-
Automated PDF report generation
-
Change tracking records
-
Timestamped calibration data
-
Environmental measurement logs
With structured QA systems like QUBYX PerfectLum, every test is stored and retrievable instantly.
When audits occur, institutions must be able to demonstrate:
-
Initial acceptance results
-
Ongoing constancy verification
-
Corrective action documentation
-
Compliance trend analysis
Audit readiness should be built into the system architecture.
6. Standardize Sensor and Measurement Protocols
Medical imaging QA best practices emphasize the importance of calibrated, traceable measurement instruments.
Best practices include:
-
Using validated photometric sensors
-
Maintaining calibration traceability
-
Ensuring sensor compatibility with QA software
-
Avoiding mixed measurement methodologies
Measurement integrity directly affects calibration accuracy. Standardized sensor workflows prevent variability across sites.
7. Monitor Ambient Light Conditions
Ambient light influences perceived contrast and grayscale discrimination.
Best practices recommend:
-
Periodic ambient light measurement
-
Defined lux thresholds per modality
-
Documentation of room lighting conditions
-
Controlled reading room policies
High ambient light can invalidate otherwise accurate DICOM calibration.
8. Integrate QA into IT and PACS Infrastructure
Medical imaging QA best practices now require integration with enterprise IT ecosystems:
-
PACS/VNA environments
-
Active Directory user management
-
Centralized compliance dashboards
-
Remote access infrastructure
QA must operate as an IT-governed system, not as an isolated physics tool.
This shift transforms display calibration into a structured compliance management system.
9. Define Clear QA Policies and Roles
Technology alone does not guarantee compliance.
Best practice requires documented policies covering:
-
Frequency of constancy testing
-
Responsibility assignments (medical physicist, IT admin, QA manager)
-
Escalation procedures for out-of-tolerance results
-
Documentation retention periods
Institutional governance ensures accountability.
10. Adopt a Lifecycle QA Strategy
Displays have a lifecycle:
-
Procurement
-
Acceptance
-
Clinical operation
-
Ongoing constancy monitoring
-
Performance degradation
-
Replacement
Medical imaging QA best practices align QA checkpoints to each lifecycle stage.
This ensures:
-
Predictable replacement planning
-
Risk reduction
-
Budget forecasting accuracy
-
Sustained diagnostic reliability
The Shift: From Isolated Calibration to Integrated QA Infrastructure
Historically, calibration was treated as a one-time adjustment. Modern medical imaging environments require:
-
Continuous performance validation
-
Centralized oversight
-
Automated scheduling
-
Audit-ready documentation
-
Enterprise governance
This is where structured QA platforms such as QUBYX PerfectLum play a critical role — enabling healthcare institutions to engineer precision rather than assume it.
Conclusion: Engineering Defensible Diagnostic Accuracy
Medical imaging QA best practices are built on five pillars:
-
Standards alignment
-
Structured acceptance testing
-
Continuous drift monitoring
-
Centralized governance
-
Audit-ready documentation
When implemented correctly, QA becomes:
-
Predictable
-
Scalable
-
Defensible
-
Compliant
In medical imaging, diagnostic confidence depends on measurable visual performance. Precision must be engineered, monitored, and documented — systematically.
Start the conversation with our calibration experts today.
In a world where every Pixel accuracy matters, PerfectLum by QUBYX proves that innovation can deliver clinical precision without financial compromise. It’s not just calibration—it’s the democratization of diagnostic imaging.
PerfectLum is Medical Display Calibration & QA Software by QUBYX LLC that delivers consistent, audit-ready display performance through standardized calibration, verification, and centralized quality assurance for radiology and teleradiology environments.
Tags:
Medical Imaging QA Best Practices, DICOM calibration, diagnostic display quality assurance, luminance drift monitoring, AAPM TG-270, DICOM GSDF, audit-ready reporting, centralized QA
Related Posts
- April 30, 2026
- News
- April 29, 2026
- News
- April 22, 2026
- News